Experiments and theory studies indicate that such tailored dual active sites realizing isolated dehydrogenation and oxidation play a crucial role in avoiding over-oxidation of ethene.Ĭatalysts preparation and characterization As a result, this strategy avoids consecutive oxidation and achieves near 100% C 2H 4 selectivity at a wider range of conversion compared with those reported in the previous work 37, 38, 39, 40. Herein, strategically tailoring dual Ni active sites is proposed by utilizing the acid sites, three-dimensional pore structure, and supercages of HY zeolite to anchor Ni 2+ Lewis acid sites (LAS) and confine NiO clusters to isolate dehydrogenation and oxidation by lattice oxygen, respectively 2, 32, 33, 34, 35, 36. Consequently, it can be feasible to avert over-oxidation that dehydrogenation and oxidation occur on different active sites. Although great effort has been made, over-oxidation remains not to be completely avoided probably due to the difficulty to manipulate identical active sites such as oxygen species responsible for both dehydrogenation and oxidation resulting in facile further oxidation of alkene on it particularly when conversion increases. It was further clarified that isolated electrophilic oxygen species played a positive role for high ethene selectivity resulted from its effective breakage of C−H bonds of ethane to selectively produce ethylene with easy desorption 30, 31. In addition, decreasing the number and restricting the local availability of active but nonselective such as electrophilic oxygen species O − could also increase alkene selectivity induced by the decrease in the probability of continuous oxidation for various metals with higher valence doped NiO materials 24, 25, 26, 27, 28, 29. Recently, hexagonal boron nitride (BN) attracted continuous interest thanks to its unexpected olefin selectivity, which originated from the stabilization of propyl species by the nitroxyl radical in the active sites of >B−O−O−N< rather than highly reactive propyl radicals thus suppressing over-oxidation 22, 23. Mo−V−Te−Nb mixed oxides were also reported to exhibit high ethene selectivity, which could be attributed to the V=O sites or redox-active O − radical sites generated by the reduction of Te 4+ to Te 0 with low reactivity to ethene 7, 8, 21. Multiple metals doped vanadium oxide, e.g. However, the activation for alkane declined 18, 19, 20. It was reported that supporting V 2O 5 and NiO with smaller clusters on the inert oxides such as SiO 2 and Al 2O 3 led to the decrease in the reactivity of active lattice oxygen species to alkene thereby its further oxidation was alleviated, which stemmed from increased metal−oxygen bond strength. Extensive studies have contributed to manipulating the chemical environment of active oxygen species to decrease its insertion as large extent as possible concurrently with activation for C−H bond in alkane. This was because alkene is facile to be further oxidized before its desorption or re-adsorbed on the active sites of dehydrogenation (usually oxygen species) according to the Mars–van Krevelen mechanism 1, 10, 11 which resulted from its higher affinity and reactivity than alkane to most surfaces particularly for those of V- and Ni-based catalysts which have been studied most for ODH 12, 13, 14, 15, 16, 17.

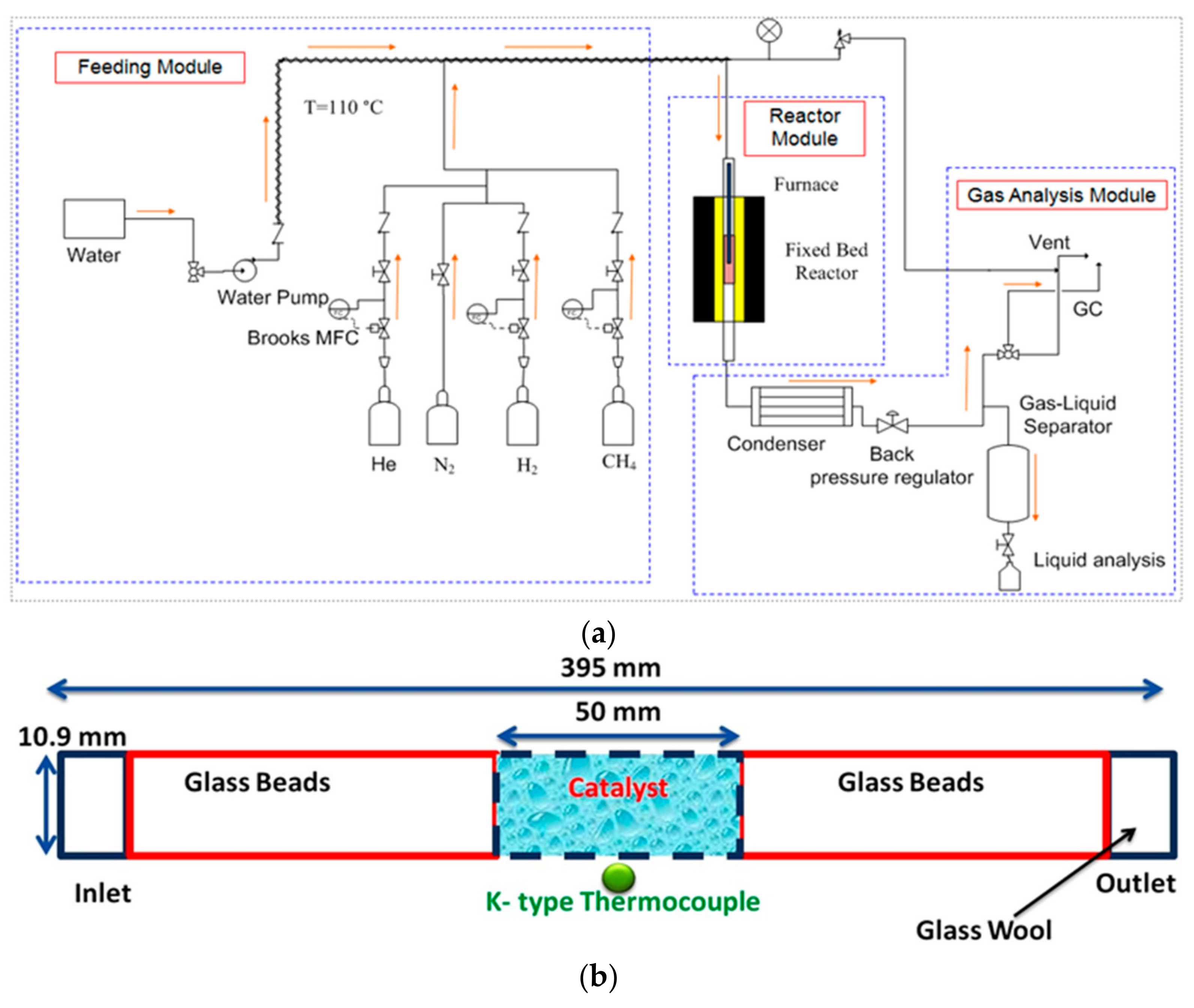
The oxidative dehydrogenation of light alkanes to olefins (ODH) which are important building blocks for a handful of industrial processes is a well-known example of such a challenging reaction that is a promising alternative to the current industrial practice of steam cracking with no thermodynamic limitation, coke formation, and large CO 2 emission but difficult to realize commercialized utilization impeded by the liable deep oxidation 6, 7, 8, 9. Hence, selectively oxidative heterogeneous catalysis to restrain over-oxidation is significant but challenging whether for fundamental studies or practical applications 2, 3, 4, 5. Unfortunately, a general and long-standing scientific conundrum is over-oxidation of desired product which is more reactive than parent substrate, impeding the industrialization process of numerous oxidation reactions with game-changing industrial value and economic benefits. Oxidation catalysis plays a pivotal role in establishing green and sustainable chemical applications and contributes approximately 30% of total production in the chemical industry, such as selective oxidation of methanol to formaldehyde, propene to acrolein, butane to maleic anhydride, and ethene to ethylene epoxide, as well as the production of key pharmaceutical intermediates 1.
